If you feel this doesn’t help you obtain a higher grade, simply email me within 7 days of purchase and I will grant a full refund.
Spectroscopy Pack
A 15-Page Summary of Spectroscopy Topics
Struggling with spectroscopy? Irritated by IR? Messed up by Mass Spec? Numbed by NMR?
This PDF collection of spectroscopy “cheat sheets” summarizes the key information you'll need to have in order to understand and solve the most commonly encountered types of spectroscopy problems. Each sheet is packed with essential detail and organized to help you understand the key concepts.
-
Immediate download for test prep -
Print ready PDF - nothing is mailed -
Contains key concepts covered in exams
What's inside
Showing 4 of 12See All
Page 1 of 12

1. Index of Unsaturation
All about calculating index of unsaturation - Hydrocarbons with no rings or multiple bonds - How double bonds and rings affect the hydrogen count - The effect of oxygen - Halogens - The unified equation
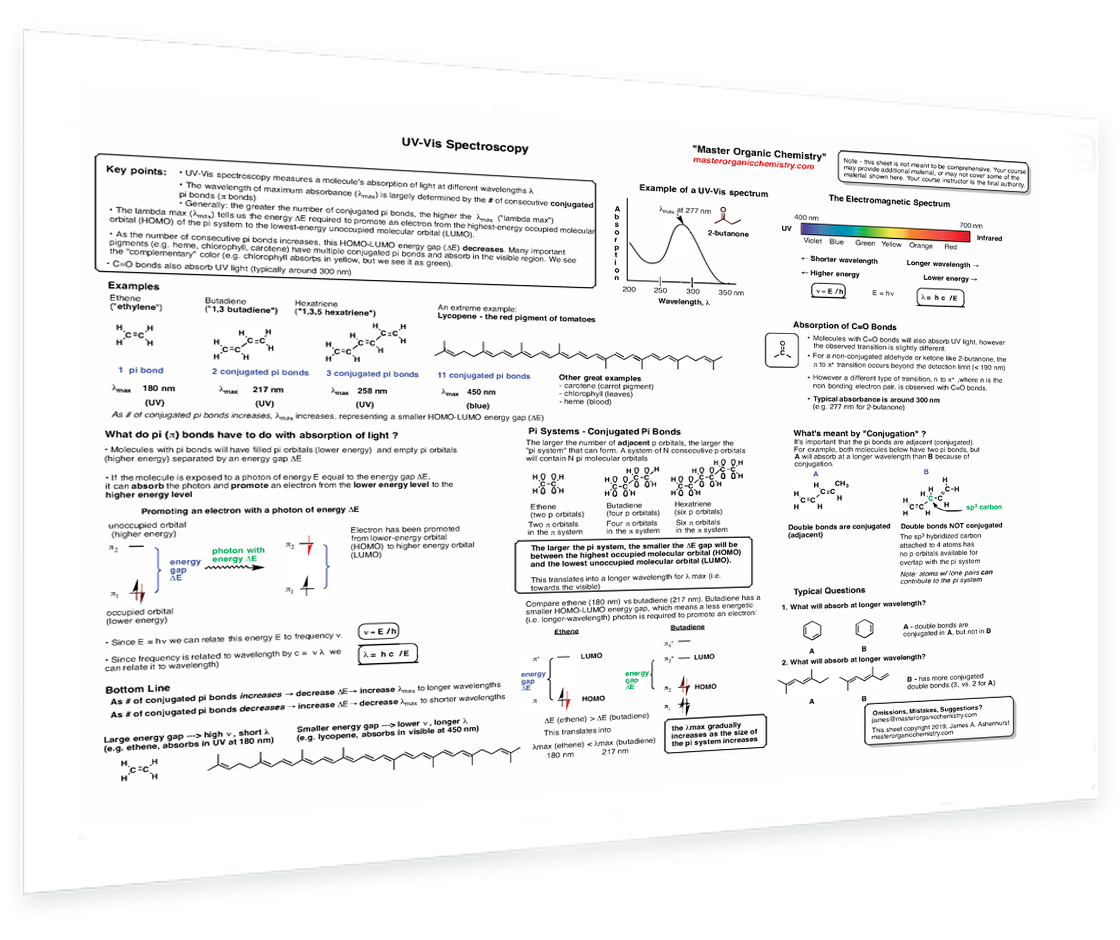
2. UV-Vis Spectroscopy
UV light and the HOMO-LUMO gap - The electromagnetic spectrum - Example of a UV-Vis spectrum - Conjugation length vs UV absorbance - Why do pi bonds absorb light? - Effect of conjugation on absorbance - Example of a UV-Vis spectrum - What absorbs at longer wavelength - Absorption of C–O bonds

3. Mass Spectrometry: Key Concepts
Mass spectrometry - Key concepts - Reading a mass spectrum - Exact mass vs molar mass - Notable isotope distributions - How to determine molecular formula from M

4. Mass Spectroscopy: Fragmentation
Mass spectrometry - Fragmentation - Understanding fragmentation - Fragment analysis - Carbocation stability trends - Examples

5. Introduction to IR Spectroscopy
Intro to IR - Key points about IR - The ball and spring analogy - Vibrational energy levels - Interpreting IR spectra - Notable IR frequencies
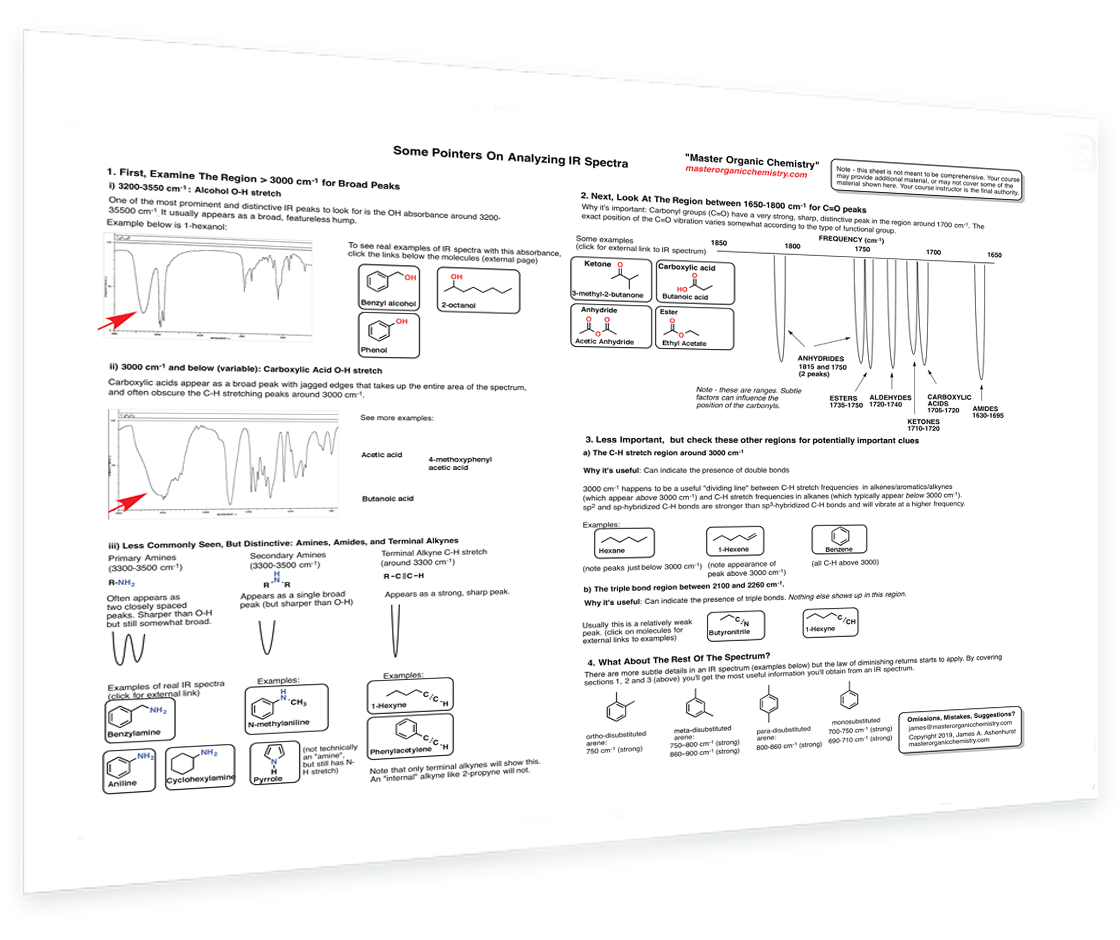
6. Pointers on Analyzing IR Spectra
Analyzing IR spectra - Alcohols - Carboxylic acids - Amines - Amides - Alkynes - Carbonyls - C-H stretch region

7. Nuclear Magnetic Resonance (NMR) Background
NMR background - Nuclear spin - Nuclei in a magnetic field - The chemical shift - What’s ppm ? - Upfield and downfield

8. NMR: How Many Signals
How to figure out "How many signals" questions - Chemical shift equivalence - Equivalence through rotation of the molecule - Equivalence through symmetry (mirror planes) - Fast rotation - Stereochemistry - Homotopic protons

9. NMR: How Many Signals (1H)
NMR - How many signals (1H) - Homotopic, Diastereotopic, and Enantiotopic protons

10. 13C NMR: Similarities & Differences
13C-NMR - Similarities and differences to 1H NMR - Chemical shift trends - Splitting in 13C NMR - Decoupled spectra

11. Understanding NMR Spectra: Chemical Shifts & Integration
Chemical shifts - Factors affecting chemical shift - Chemical shifts of common C-H bonds - Integration - Splitting

12. NMR: Splitting
Chemical shifts and integration - Chemical shift - The role of electron withdrawing groups - The area under signal is proportional to the number of protons - Dealing with integration curves
Your purchase is safe
- James Ashenhurst, Owner






